
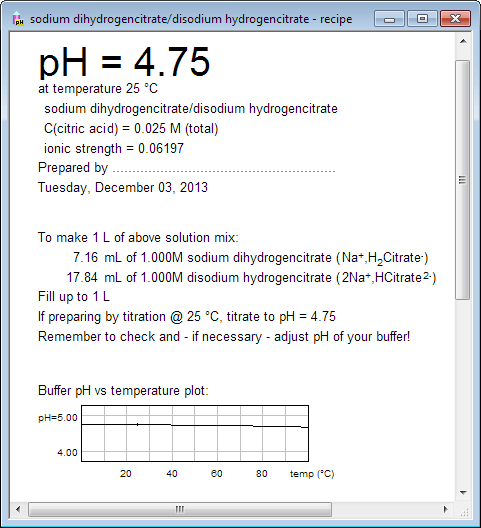
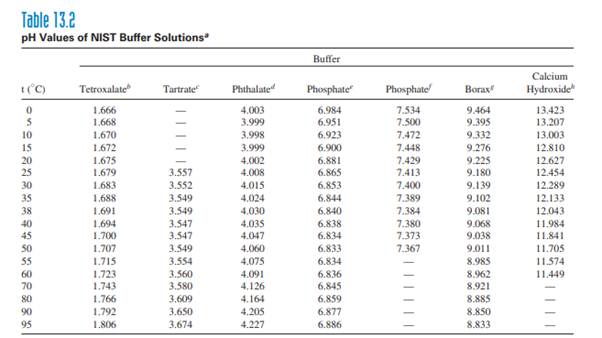
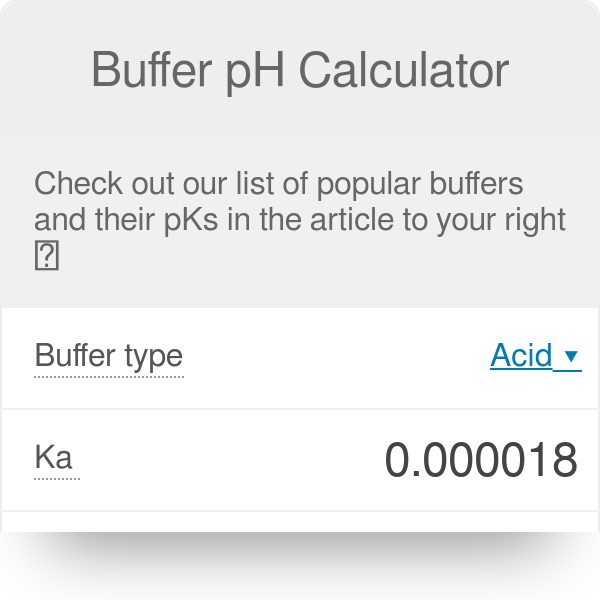
SOLVED: A phosphate buffer solution is prepared by dissolving 25.0 g of potassium hydrogen phosphate (K2HPO4; FM = 174.18) and 12.0 g of potassium dihydrogen phosphate (KH2PO4; FM = 136.09) in 250
The Preparation of Acetate and Phosphate Buffer Solutions of Known PH and Ionic Strength | Journal of the American Chemical Society


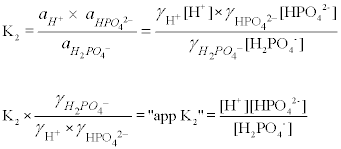




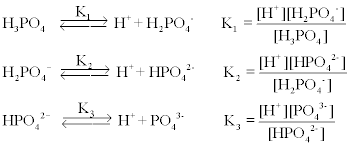


![BS021] 0.5M Sodium Phosphate Buffer, pH x.x | Biosolution BS021] 0.5M Sodium Phosphate Buffer, pH x.x | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2017/11/BS020-Sodium-Phosphate-Buffer.jpg)





