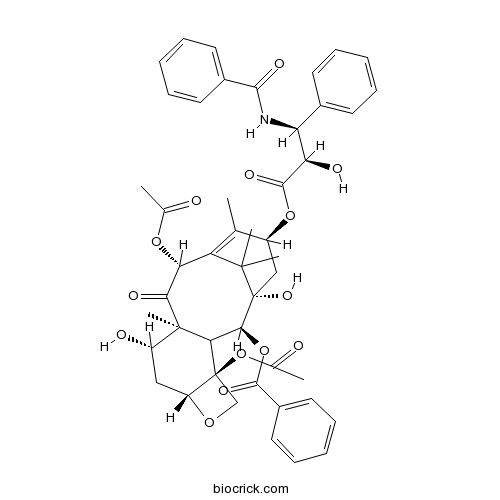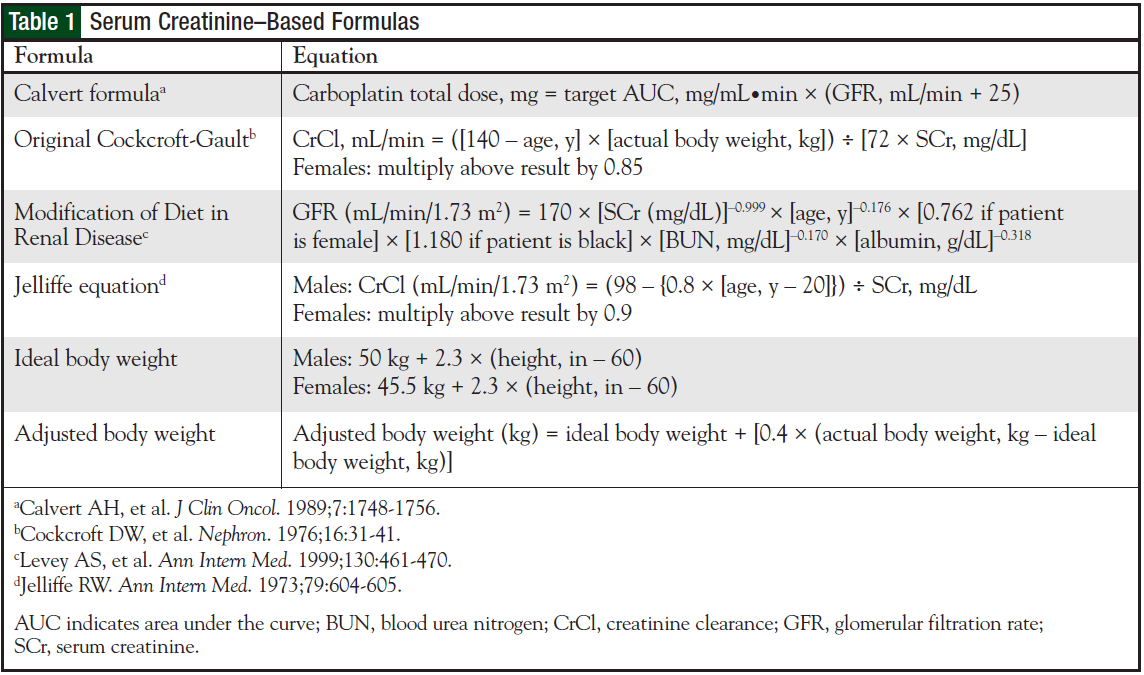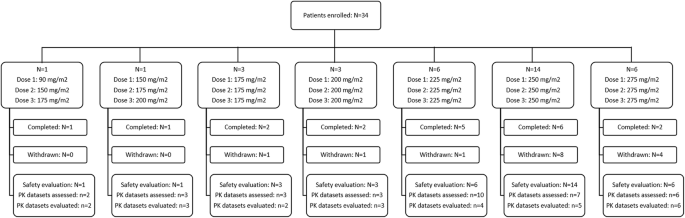
Maximum Tolerated Dose and Pharmacokinetics of Paclitaxel Micellar in Patients with Recurrent Malignant Solid Tumours: A Dose-Escalation Study | SpringerLink
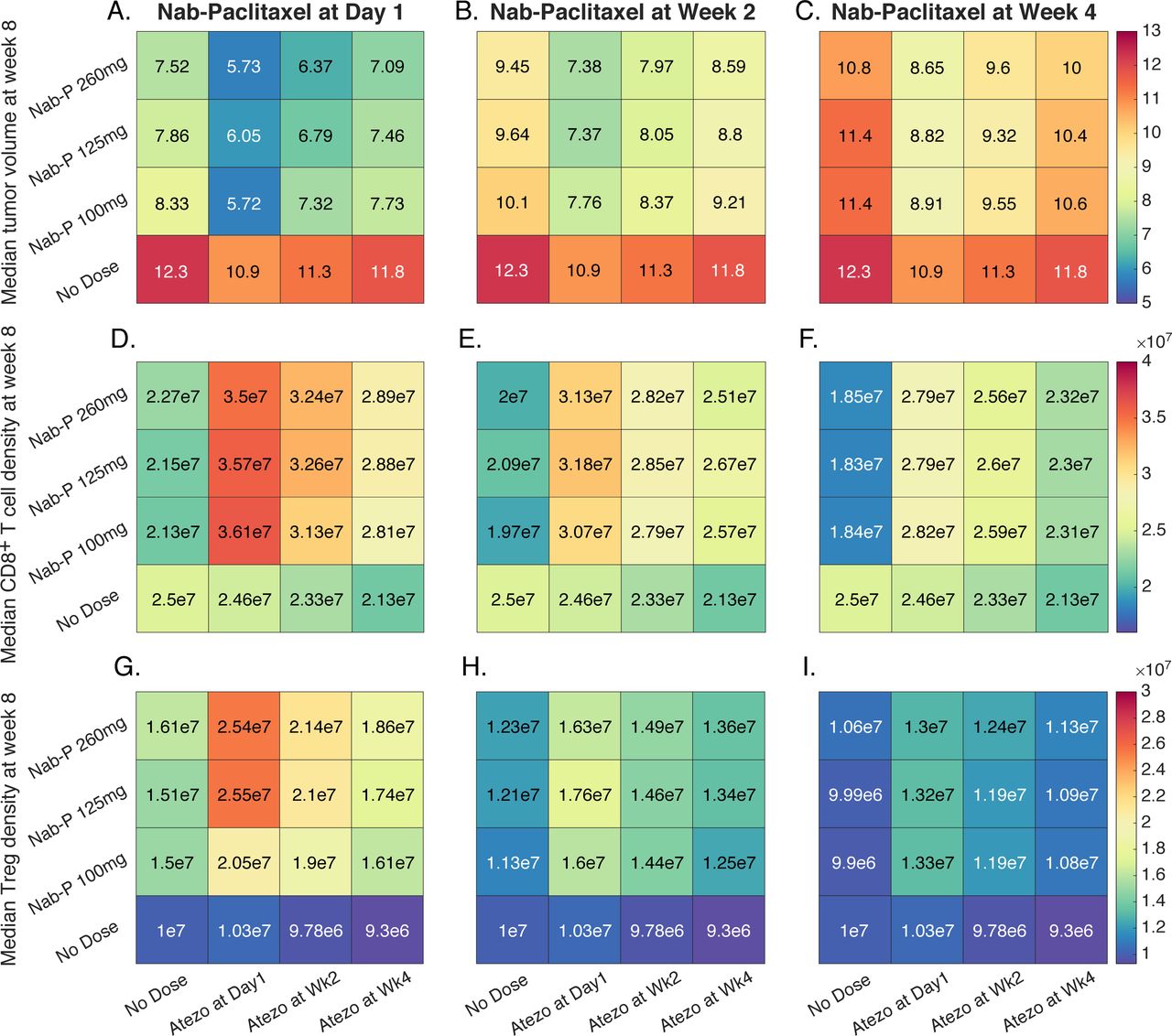
Quantitative systems pharmacology model predictions for efficacy of atezolizumab and nab-paclitaxel in triple-negative breast cancer | Journal for ImmunoTherapy of Cancer

A Dose Finding Study of Weekly and Every-3-Week nab-Paclitaxel Followed by Carboplatin as First-Line Therapy in Patients with Advanced Non-small Cell Lung Cancer - ScienceDirect

Open-label, randomized study of individualized, pharmacokinetically (PK)-guided dosing of paclitaxel combined with carboplatin or cisplatin in patients with advanced non-small-cell lung cancer (NSCLC)† - Annals of Oncology

Preclinical tolerance evaluation of the addition of a cisplatin-based dry powder for inhalation to the conventional carboplatin-paclitaxel doublet for treatment of non-small cell lung cancer - ScienceDirect
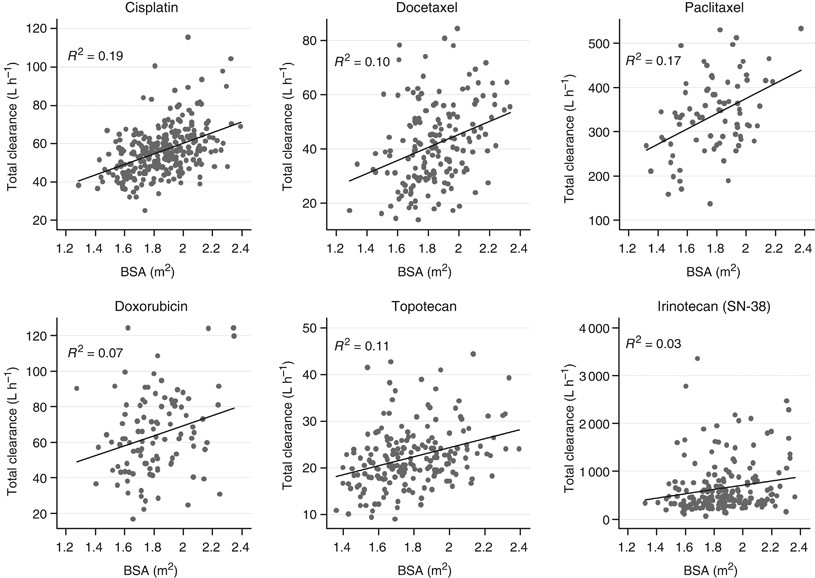
Dose banding as an alternative to body surface area-based dosing of chemotherapeutic agents | British Journal of Cancer

Randomized study of individualized pharmacokinetically‐guided dosing of paclitaxel compared with body‐surface area dosing in Chinese patients with advanced non‐small cell lung cancer - Zhang - 2019 - British Journal of Clinical Pharmacology -

Effect of body mass index on pharmacokinetics of paclitaxel in patients with early breast cancer - Gota - 2021 - Cancer Medicine - Wiley Online Library