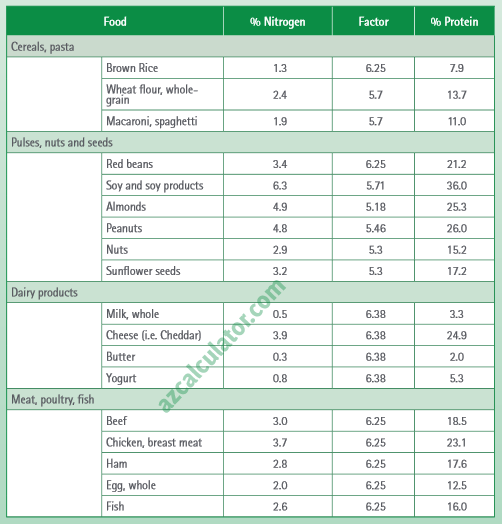
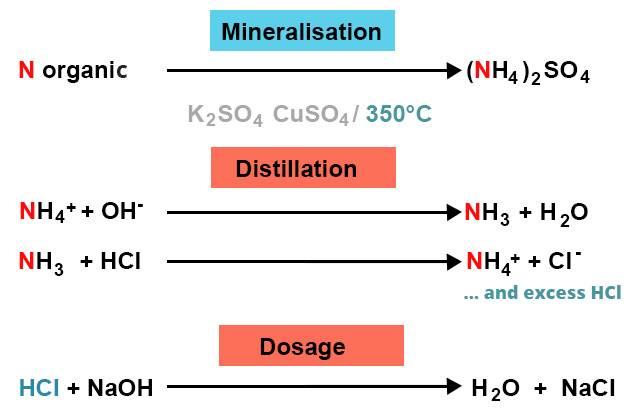
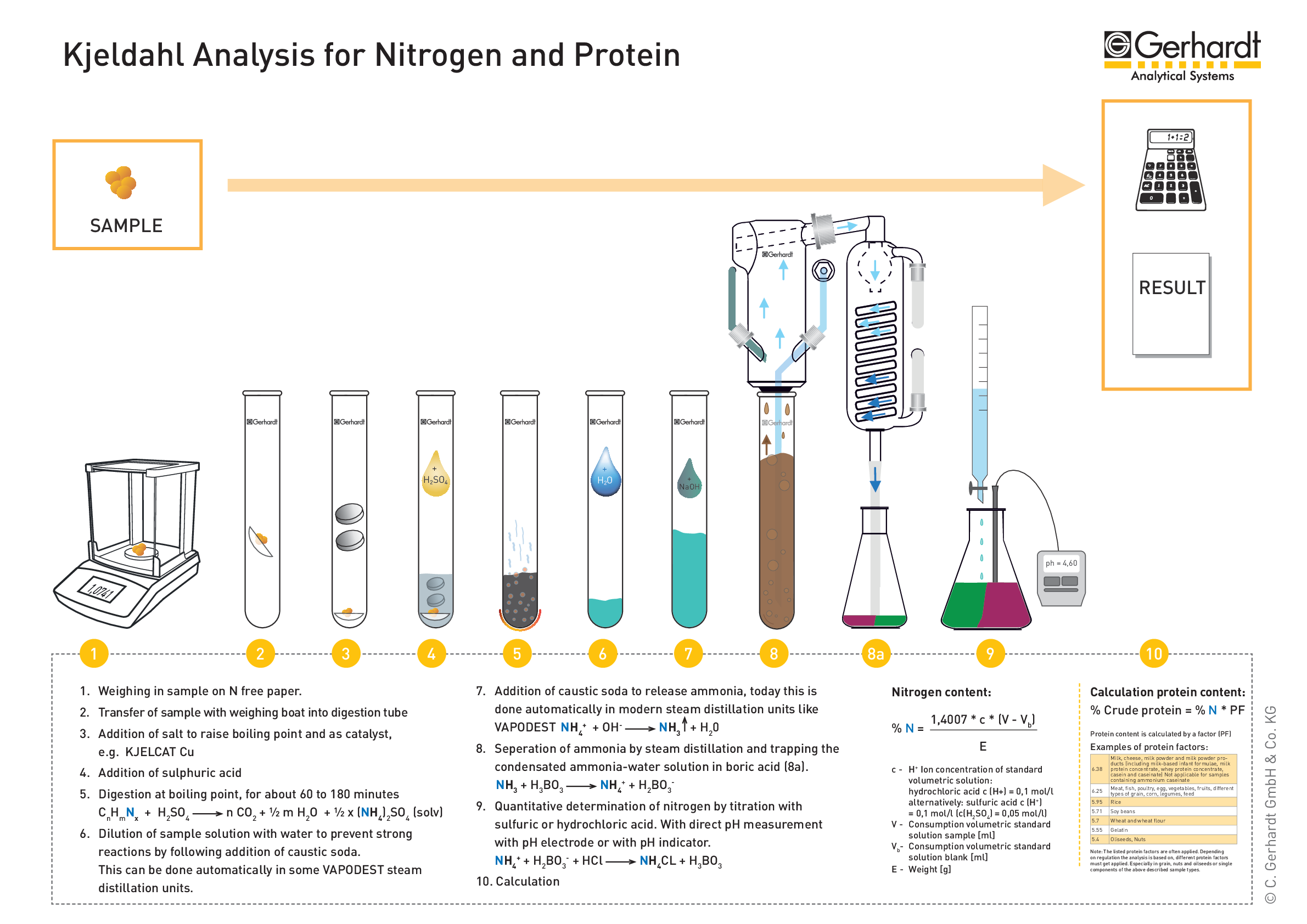
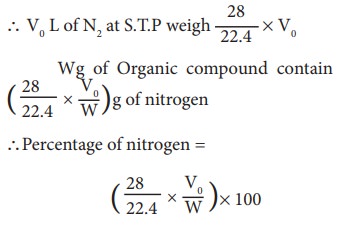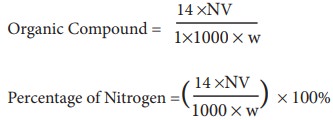In the Kjedahl's method for estimation of nitrogen present in soil sample, ammonia evolved from 0.75 g of sample neutralized 10ml. of 1M H2SO4 . The percentage of nitrogen in the soil is:

PDF) Comparison of the Kjeldahl method, Dumas method and NIR method for total nitrogen determination in meat and meat products

In the estimation of nitrogen by Kjeldahl's method, 2.8 g of an organic compound required 20 millimoles of H(2)SO(4) for the complete neutralisation of NH(3) gas evolved. The percentage of nitrogen in

In the Kjedahl's method for estimation of nitrogen present in soil sample, ammonia evolved from 0.75 g of sample neutralized 10ml. of 1M H2SO4 . The percentage of nitrogen in the soil is:

SOLVED: The Kjeldahl method is commonly used to determine the percent protein in meat products. The majority of protein molecules in living matter contain just about 16% nitrogen (N). A 1.250 g

In the Kjedahl's method for estimation of nitrogen present in soil sample, ammonia evolved from 0.75 g of sample neutralized 10ml. of 1M H2SO4 . The percentage of nitrogen in the soil is:
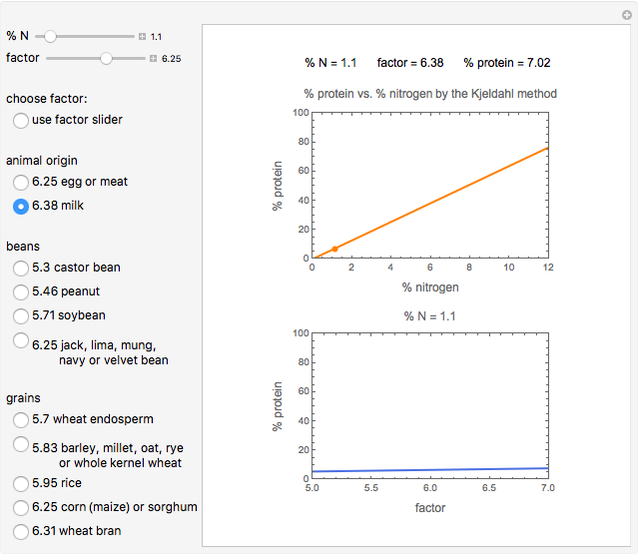
Kjeldahl Method for Determining Percent Protein from Percent Nitrogen - Wolfram Demonstrations Project