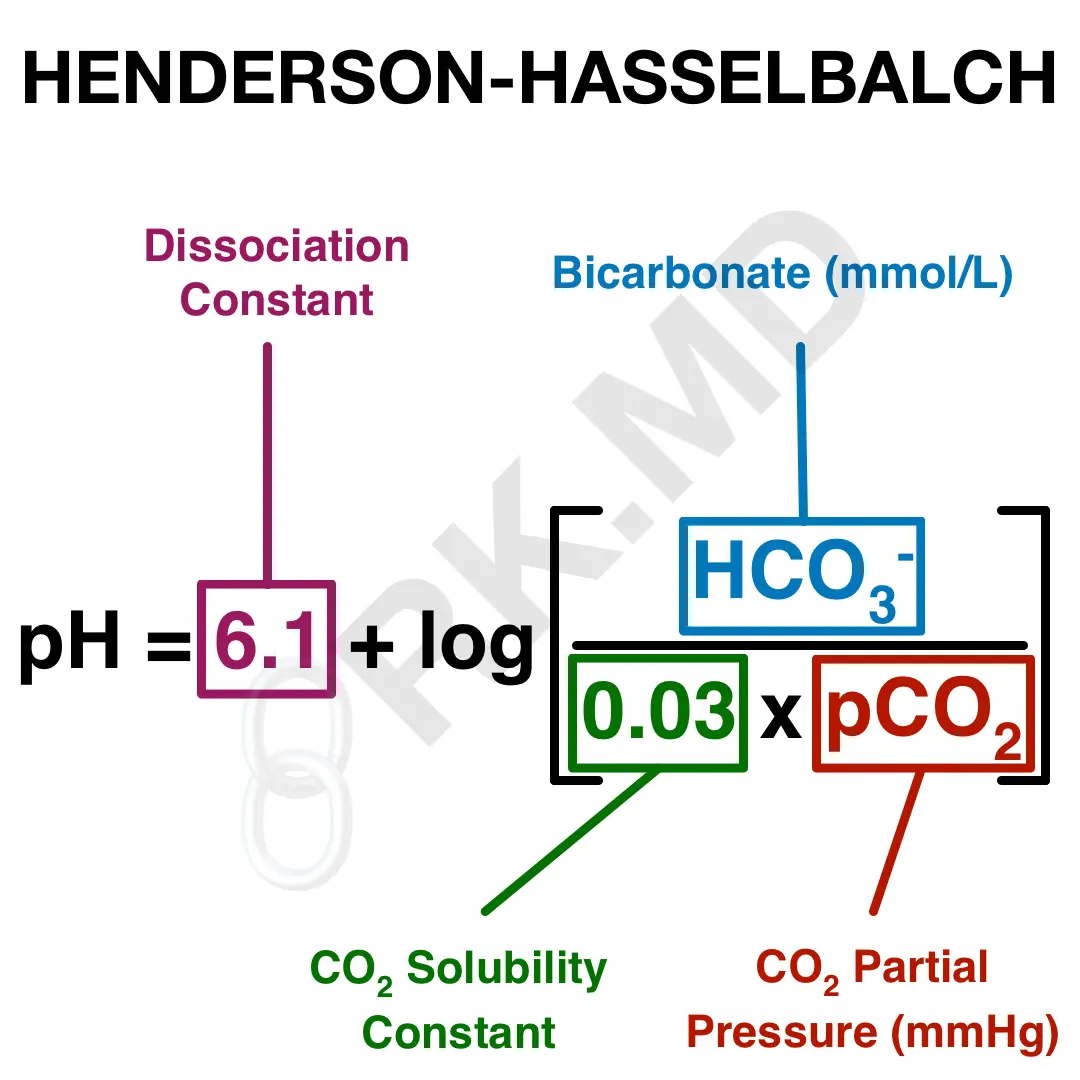
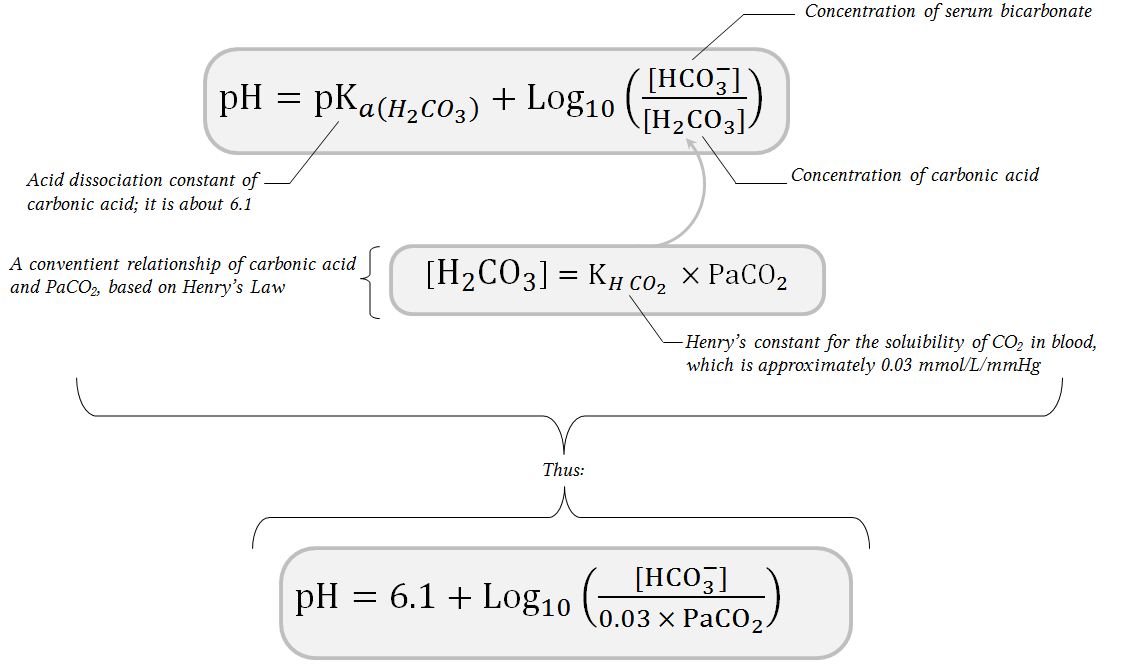
SOLVED: The pH of human blood is maintained at7.40 and is primarily controlled by the carbonic acid/bicarbonate (HCO/HCO)acid-base system.Use the HH equation to calculation the HCO3concentration if the HCO3 concentration is 24.5

Can someone explain where did the CO3(2-) and HCO3(-) came from? And how do I write the equations with HCl and Na2CO3? : r/chemhelp

Calculated arterial blood bicarbonate (HCO3) and partial pressure of... | Download Scientific Diagram

Bicarbonate anion ( HCO3 ) - structural chemical formula and molecule model. Vector illustration Stock Vector Image & Art - Alamy
![PDF] Discrepancy between Measured Serum Total Carbon Dioxide Content and Bicarbonate Concentration Calculated from Arterial Blood Gases | Semantic Scholar PDF] Discrepancy between Measured Serum Total Carbon Dioxide Content and Bicarbonate Concentration Calculated from Arterial Blood Gases | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d604591e54c0e843b180644b6e556990864a0b37/7-Figure4-1.png)
PDF] Discrepancy between Measured Serum Total Carbon Dioxide Content and Bicarbonate Concentration Calculated from Arterial Blood Gases | Semantic Scholar


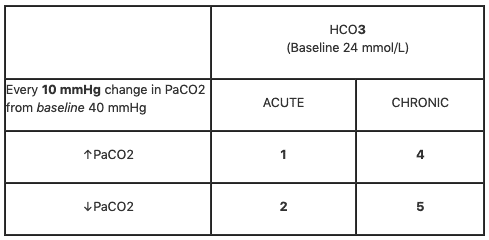


![Solved Question 2 Calculate the blood [HCO3] of a patient | Chegg.com Solved Question 2 Calculate the blood [HCO3] of a patient | Chegg.com](https://media.cheggcdn.com/study/965/965de12f-e609-4ec1-80e3-f55348aade41/image)



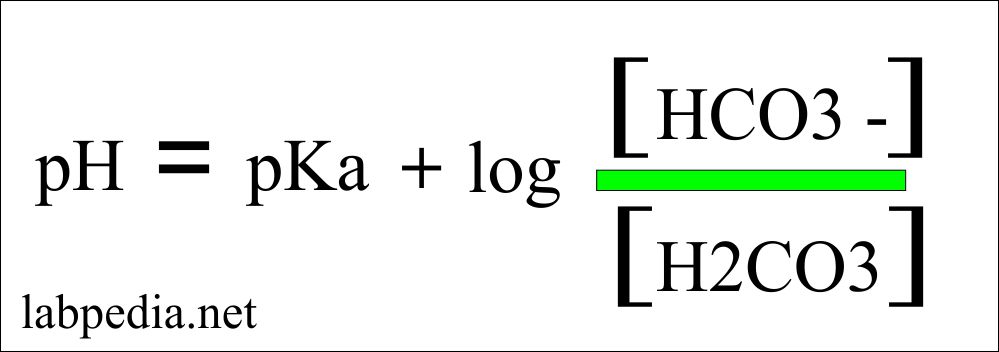
![Solved 3. calculate [H^+] based on the pH measurement in | Chegg.com Solved 3. calculate [H^+] based on the pH measurement in | Chegg.com](https://media.cheggcdn.com/study/973/9738f4a5-a8d9-4722-8a34-990ac1ca6db2/image.png)